At the forefront of cancer drug development and treatment design
Using state-of-the-art experimental approaches in genomics, genetics, molecular biology, cell biology, imaging, biochemistry, and animal modeling, the Department of Pharmacology & Therapeutics at Roswell Park is making breakthrough discoveries in new cancer therapies.
What we do
We are hiring faculty positions at the Assistant Professor level in the Department of Pharmacology and Therapeutics.
Learn more- Identify novel molecular targets for therapeutic development
- Characterize the mechanisms of action and preclinical development of novel drug candidates
- Clinically translate in-house developed anti-cancer therapeutics
- Develop mechanism-driven novel combination therapies
Our researchers collaborate extensively with each other, with other scientists at Roswell Park and elsewhere, and with partners in the pharmaceutical industry, to leverage expertise from a variety of disciplines.
We also participate in Roswell Park’s CCSG Developmental Therapeutics (DT) Program and Translational Research Groups (TRG) for specific cancer types, and the University at Buffalo (UB) graduate training programs — mainly, the Experimental Therapeutics (ET) program.
Research highlights
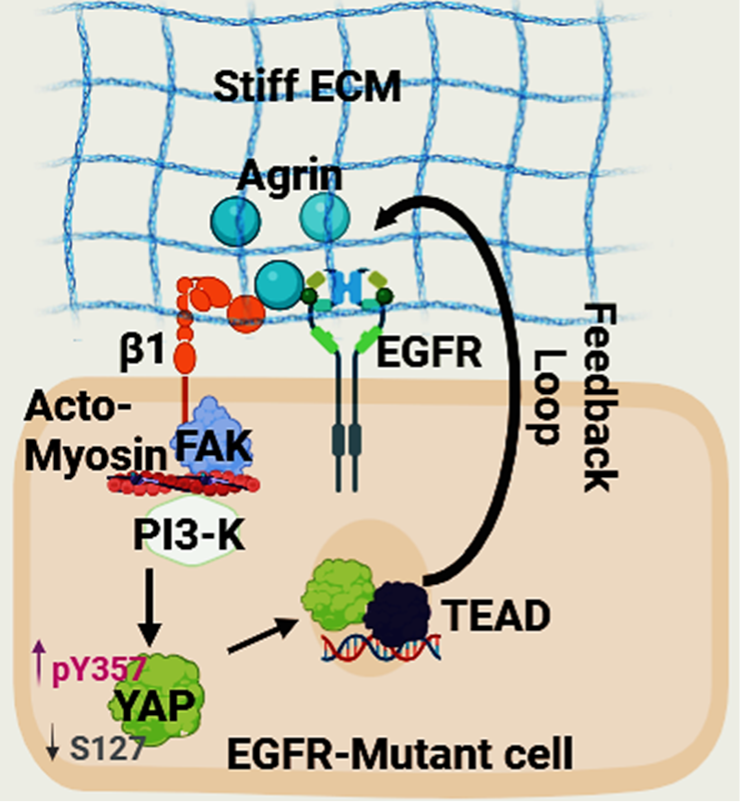
An Agrin–YAP/TAZ Rigidity Sensing Module Drives EGFR-Addicted Lung Tumorgenesis
Advanced Science
Principal Investigator: Dr. Sayan Chakraborty
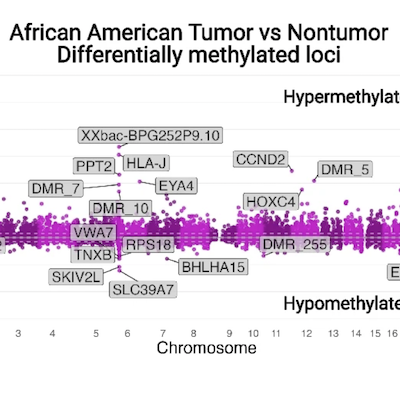
Race-specific coregulatory and transcriptomic profiles associated with DNA methylation and androgen receptor in prostate cancer
Genome Medicine
Principal Investigator: Dr. Anna Woloszynska
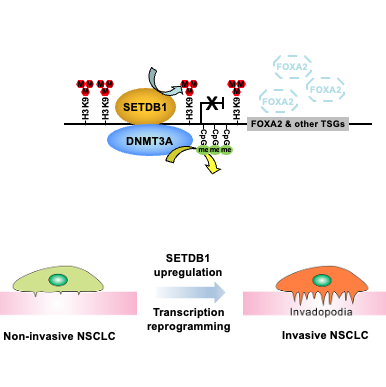
Histone H3K9 methyltransferase SETDB1 augments invadopodia formation to promote tumor metastasis
Oncogene
Principal Investigator: Dr. Jia Fang
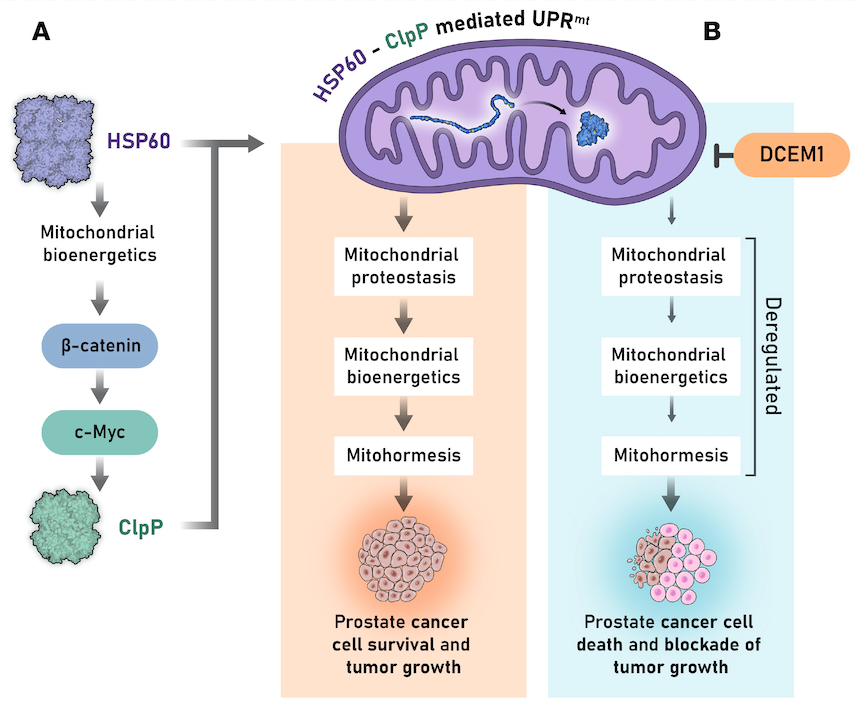
A mitochondrial unfolded protein response inhibitor suppresses prostate cancer growth in mice via HSP60
Journal of Clinical Investigation
Principal Investigator: Dr. Dhyan Chandra
Intron retention is a hallmark and spliceosome represents a therapeutic vulnerability in aggressive prostate cancer
Nature Communications
Principal Investigator: Dr. Dean Tang
Kirk JS, Wang J, Long M, Rosario S, Tracz A, Ji Y, Kumar R, Liu X, Jamroze A, Singh PK, Puzanov I, Chatta G, Cheng Q, Huang J, Wrana JL, Lovell J, Yu H, Liu S, Shen MM, Liu T, Tang DG. Integrated single-cell analysis defines the epigenetic basis of castration-resistant prostate luminal cells. Cell Stem Cell. 2024 Jun 14:S1934-5909(24)00185-1. doi: 10.1016/j.stem.2024.05.008. Online ahead of print.PMID: 38878775
Alahmari AA, Chaubey AH, Jonnakuti VS, Tisdale AA, Schwarz CD, Cornwell AC, Maraszek KE, Paterson EJ, Kim M, Venkat S, Cortes Gomez E, Wang J, Gurova KV, Yalamanchili HK, Feigin ME. CPSF3 inhibition blocks pancreatic cancer cell proliferation through disruption of core histone mRNA processing. RNA. 2024 Jan 8:rna.079931.123. doi: 10.1261/rna.079931.123. Epub ahead of print. PMID: 38191171.
Doshi, B., Athans, S.R. & Woloszynska, A. Biological differences underlying sex and gender disparities in bladder cancer: current synopsis and future directions. Oncogenesis 12, 44 (2023). https://doi.org/10.1038/s41389-023-00489-9
Ueshima S, Fang J. Histone H3K9 methyltransferase SETDB1 augments invadopodia formation to promote tumor metastasis. Oncogene. 2022 Jun;41(24):3370-3380. doi: 10.1038/s41388-022-02345-3. Epub 2022 May 11. PubMed PMID: 35546351; PubMed Central PMCID: PMC9801494.
Inigo J, Kumar R, Chandra D. Targeting the mitochondrial unfolded protein response in cancer: opportunities and challenges. Inigo, Joseph R. et al. Trends in Cancer, Volume 7, Issue 12, 1050 - 1053
Venkat S, Alahmari A, Feigin M. Drivers of Gene Expression Dysregulation in Pancreatic Cancer. Venkat, Swati et al. Trends in Cancer, Volume 7, Issue 7, 594 - 605
Li F, Aljahdali I, Zhang R, Nastiuk K, Krolewski J, Ling X. Kidney cancer biomarkers and targets for therapeutics: survivin (BIRC5), XIAP, MCL-1, HIF1, HIF2, NRF2, MDM2, MDM4, p53, KRAS and AKT in renal cell carcinoma. J Exp Clin Cancer Res 40, 254 (2021).
Zhang Y. The root cause of drug resistance in HER2-positive breast cancer and the therapeutic approaches to overcoming the resistance. Pharmacology & Therapeutics, Volume 218, 2021, 107677, ISSN 0163-7258,
O'Malley J, Kumar R, Inigo J, Yadav N, Chandra D. Mitochondrial Stress Response and Cancer. Trends in Cancer, Volume 6, Issue 8, 2020, Pages 688-701, ISSN 2405-8033.
Venkat S, Tisdale A, Schwarz JR, Alahmari A, Maurer HC, Olive KP, Eng K, Feigin M. Alternative polyadenylation drives oncogenic gene expression in pancreatic ductal adenocarcinoma. Genome Res. 2020 Mar;30(3):347-360. doi: 10.1101/gr.257550.119. Epub 2020 Feb 6.
Cornwell A, Feigin M. Unintended Effects of GPCR-Targeted Drugs on the Cancer Phenotype. Cornwell, Abigail C. et al. Trends in Pharmacological Sciences, Volume 41, Issue 12, 1006 - 1022
Zhang D, Hu Q, Liu X, Ji Y, Chao HP, Liu Y, Tracz A, Kirk J, Buonamici S, Zhu P, Wang J, Liu S, Tang D. Intron retention is a hallmark and spliceosome represents a therapeutic vulnerability in aggressive prostate. Zhang, D., Hu, Q., Liu, X. et al. Intron retention is a hallmark and spliceosome represents a therapeutic vulnerability in aggressive prostate cancer. Nat Commun 11, 2089 (2020).
Kumar R, Bhat T, Walsh EM, Chaudhary A, O'Malley J, Rhim JS, Wang J, Morrison C, Attwood K, Bshara W, Mohler J, Yadav N, Chandra D. Cytochrome c Deficiency Confers Apoptosome and Mitochondrial Dysfunction in African-American Men with Prostate Cancer. Cancer Res 1 April 2019; 79 (7): 1353–1368.
Mukhopadhyay UK, Oturkar C, Adams C, Wickramasekera N, Bansal S, Medisetty R, Miller A, Swetzig WM, Silwal-Pandit L, Borresen-Dale AL, Creighton CJ, Park JH, Konduri SD, Mukhopadhyay A, Caradori A, Omilian A, Bshara W, Kaipparettu BA, Das G. TP53 Status as a Determinant of Pro- vs Anti-Tumorigenic Effects of Estrogen Receptor-Beta in Breast Cancer. JNCI: Journal of the National Cancer Institute, Volume 111, Issue 11, November 2019, Pages 1202–1215,
Knudsen E, Pruitt S, Hershberger P, Witkiewicz A, Goodrich D. Cell Cycle and Beyond: Exploiting New RB1 Controlled Mechanisms for Cancer Therapy. Knudsen, Erik S. et al. Trends in Cancer, Volume 5, Issue 5, 308 - 324
Ramakrishnan S, Granger V, Rak M, Hu Q, Attwood K, Aquila L, Krishnan N, Osiecki R, Azabdaftari G, Guru K, Chatta G, Gueron G, McNally L, Ohm J, Wang J, Woloszynska A. Inhibition of EZH2 induces NK cell-mediated differentiation and death in muscle-invasive bladder cancer. Cell Death Differ 26, 2100–2114 (2019).
Yang L, Li Y, Bhattacharya A, Zhang Y. A recombinant human protein targeting HER2 overcomes drug resistance in HER2-positive breast cancer. Pharmacology & Therapeutics, 218, (107677), (2021).
Li Q, Liu B, Chao HP, Ji Y, Lu Y, Mehmood R, Jeter C, Chen T, Moore JR, Li W, Liu C, Rycaj K, Tracz A, Kirk J, Calhoun-Davis T, Xiong J, Deng Q, Huang J, Foster B, Gokhale A, Chen X, Tang D. LRIG1 is a pleiotropic androgen receptor-regulated feedback tumor suppressor in prostate cancer. Nat Commun 10, 5494 (2019).
Kumar S, Inigo J, Kumar R, Chaudhary A, O'Malley J, Balachandar S, Wang J, Attwood K, Yadav N, Hochwald S, Wang X, Chandra D. Nimbolide reduces CD44 positive cell population and induces mitochondrial apoptosis in pancreatic cancer cells. Cancer Letters, Volume 413, 2018, Pages 82-93, ISSN 0304-3835, doi: 10.1016/j.canlet.2017.10.029.
Li Q, Deng Q, Chao HP, Liu X, Lu Y, Lin K, Liu B, Tang GW, Zhang D, Tracz A, Jeter C, Rycaj K, Calhoun-Davis T, Huang J, Rubin MA, Beltran H, Shen J, Chatta G, Puzanov I, Mohler J, Wang J, Zhao R, Kirk J, Chen X, Tang D. Linking prostate cancer cell AR heterogeneity to distinct castration and enzalutamide responses. Nat Commun 9, 3600 (2018).
Zhang D, Zhao S, Li X, Kirk J, Tang D. Prostate Luminal Progenitor Cells in Development and Cancer. Zhang, Dingxiao et al. Trends in Cancer, Volume 4, Issue 11, 769 - 783.
Feigin M, Garvin T, Bailey P, Waddell N, Chang DK, Kelley DR, Shuai S, Gallinger S, McPherson JD, Grimmond SM, Khurana E, Stein LD, Biankin AV, Schatz MC, Tuveson DA. Recurrent noncoding regulatory mutations in pancreatic ductal adenocarcinoma. Nat Genet 49, 825–833 (2017).
Ku S, Rosario S, Wang Y, Mu P, Seshadri M, Goodrich Z, Goodrich M, Labbe D, Gomez E, Wang J, Long H, Xu B, Brown M, Loda M, Sawyers C, Ellis L, Goodrich D. Rb1 and Trp53 cooperate to suppress prostate cancer lineage plasticity, metastasis, and antiandrogen resistance. Science (2017), Vol 355, Issue 6320 pp. 78-83
Yang L, Li Y, Bhattacharya A, Zhang Y. PEPD is a pivotal regulator of p53 tumor suppressor. Nat Commun 8, 2052 (2017).
Liu C, Liu N, Zhang D, Deng Q, Liu B, Chao HP, Rycaj K, Takata Y, Lin K, Lu Y, Zhong Y, Krolewski J, Shen J, Tang D. MicroRNA-141 suppresses prostate cancer stem cells and metastasis by targeting a cohort of pro-metastasis genes. Nat Commun 8, 14270 (2017).
Atanassov B, Mohan RD, Lan X, Kuang X, Lu Y, Lin K, McIvor E, Li W, Zhang Y, Florens L, Byrum SD, Mackintosh SG, Calhoun-Davis T, Koutelou E, Wang L, Tang D, Tackett AJ, Washburn MP, Workman JL, Dent SYR. ATXN7L3 and ENY2 Coordinate Activity of Multiple H2B Deubiquitinases Important for Cellular Proliferation and Tumor Growth. Atanassov, Boyko S. et al. Molecular Cell, Volume 62, Issue 4, 558 - 571.
Zhang D, Park D, Zhong Y, Lu Y, Rycaj K, Gong S, Chen X, Liu X, Chao H-P, Whitney P, Calhoun-Davis T, Takata Y, Shen J, Iyer VR, Tang D. Stem cell and neurogenic gene-expression profiles link prostate basal cells to aggressive prostate cancer. Nat Commun 7, 10798 (2016).
Chen X, Li Q, Liu X, Liu C, Liu R, Rycaj K, Zhang D, Liu B, Jeter C, Calhoun-Davis T, Lin K, Lu Y, Chao HP, Shen J, Tang D. Defining a Population of Stem-like Human Prostate Cancer Cells That Can Generate and Propagate Castration-Resistant Prostate Cancer. Clin Cancer Res 1 September 2016; 22 (17): 4505–4516.
Training the next generation of scientists
In addition to our research and community outreach, the Department of Pharmacology & Therapeutics is actively involved in hands-on teaching with Master’s and PhD students in our highly competitive graduate studies program.
Our multidisciplinary education model allows students a unique immersive experience, preparing them for successful careers as independent investigators.
Hannah Calkins (Hershberger Lab) won the People’s Choice Award in the 2022 Three Minute Thesis competition at the University at Buffalo. Her presentation was entitled, “Combating Treatment Resistance in Lung Cancer.”
Sarah Athans (Woloszynska Lab) presented her thesis work in a poster presentation at the 2022 AACR Annual Meeting in New Orleans.
Abby Cornwell (Feigin Lab) was awarded an F31 Ruth L. Kirschstein Predoctoral Individual Fellowship from the National Institutes of Health.
Recent grant funding
Herd of Hope Team Science Award, Roswell Park Alliance Foundation
Developing and Translating Innovative Strategies to Therapeutically Target Prostate Cancer Lineage Plasticity
Principal investigators: Dean Tang, David Goodrich, Gurkamal Chatta, Dominic Smiraglia
Meet Dr. TangNational Institutes of Health
The role of USP27X-Cyclin D1 axis in HER2 Therapy Resistant Breast Cancer
Principal Investigator: Boyko Atanassov
Meet Dr. AtanassovAtanassov LabNational Institutes of Heatlh
Dissecting the Mechanism of SETDB1 and its K867 Monoubiquitination in Lung Cancer Progression
Principal Investigator: Jia Fang
Meet Dr. FangDepartment of Defense
Department of Defense Slow-Cycling Prostate Cancer Cells in Therapy Resistance, Disease Relapse and Metastasis
Principal Investigator: Dean Tang
Meet Dr. TangProstate Cancer Foundation
Developing and Translating Innovative Strategies to Therapeutically Target Prostate Cancer Lineage Plasticity
Principal Investigators: David Goodrich, Gurkamal Chatta, Dean Tang, Dominic Smiraglia, Mark Long
Meet Dr. GoodrichGoodrich LabSpotlight: Developmental Therapeutics Research Program
How can we use new treatments to target cancer cells and reduce the body’s resistance? Scientists across Roswell Park are studying how to develop and translate novel mechanism-driven anti-cancer therapeutics and therapeutic combinations.
Our goal is, by employing state-of-the-art technologies and the most updated knowledge, to develop novel mechanism-driven anticancer therapeutics and therapeutic combinations.
Dean Tang, PhD
In the news
Contact us
Susan Bechtel
Department Administrator
Phone: 716-845-5853
Fax: 716-845-8857
Email: Susan.Bechtel@RoswellPark.org