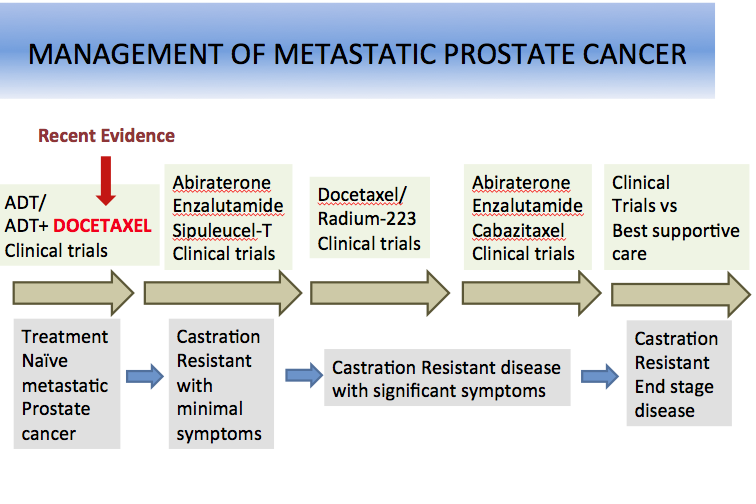
The traditional algorithm of management for metastatic prostate cancer involves androgen deprivation therapy (ADT) as first-line treatment with reservation of chemotherapy for second-line setting after disease progression. However, A phase III study led by the National Cancer Institute and recently presented at the American Society of Clinical Oncology (ASCO) 2014 annual meeting demonstrated therapeutic benefit of adding docetaxel to ADT in the frontline setting.
A total of 790 men with newly diagnosed metastatic prostate cancer were randomized in a 1:1 fashion to ADT alone (N=393) or docetaxel (75 mg/m2 every 3 weeks for maximum 6 cycles) in combination with ADT (N=397). Chemotherapy was initiated within 4 months of androgen deprivation therapy. Median age was 63 years, ECOG performance status was 0 or 1 in 98% of patients, 24% of patients had received prior radiation therapy, and 24% had undergone prostatectomy. Majority of patients had high-volume disease (64% in ADT alone arm and 67% in combination arm). At a median follow-up of 29 months, with 137 deaths in ADT alone arm vs 104 deaths in combination arm, an improvement of 13 months in overall survival (OS) was seen with addition of docetaxel (median OS of 44 months in ADT alone arm and 57.6 months in combination arm).
The magnitude of impact on median OS was considerably higher in the subset of 520 patients with high-volume disease (32.2 months in ADT alone vs 49.2 months in combination). Additional parameters such as delay in disease progression assessed by PSA suppression (PSA < 0.2 ng/mL at one year: 11.7% vs 22.7% in favor of combination arm), appearance of new metastases or symptoms (19.8 months vs 32.7 months in favor of combination arm) also supported utilization of combination therapy. Toxicity assessment revealed neutropenic fever in 6% and grade 3 neuropathy with sensory and motor dysfunction in 1% each in the combination arm, as well as one treatment-related death in combination arm. The authors concluded that combination chemotherapy is more effective, but is associated with an adverse effect profile warranting careful selection of patients, especially favoring patients with high burden of disease (skeletal and or visceral). Data for patients with low burden of disease are not conclusive and require longer follow-up.
Multiple ongoing studies at Roswell Park Comprehensive Cancer Center are evaluating additional novel approaches for management of prostate cancer. Click here to view all available clinic studies.