Dysregulation of hematopoiesis in immunotherapy
Research in the lab of Michael Nemeth, PhD, centers on understanding the mechanisms by which dysregulation of blood cell development – also known as “hematopoiesis” – in the context of cancer results in an immunologic milieu that is unable to support successful immunotherapy.
Dr. Nemeth’s team is particularly interested in studying this problem in patients with blood cell cancers such as myelodysplastic syndromes (MDS) and in patients with solid tumors such as breast cancer.
Research focus: Myelodysplastic syndromes
Our experience with immunotherapy in myeloid cancers suggests that understanding of immunologic milieu in these diseases is required to maximize the response to therapy.
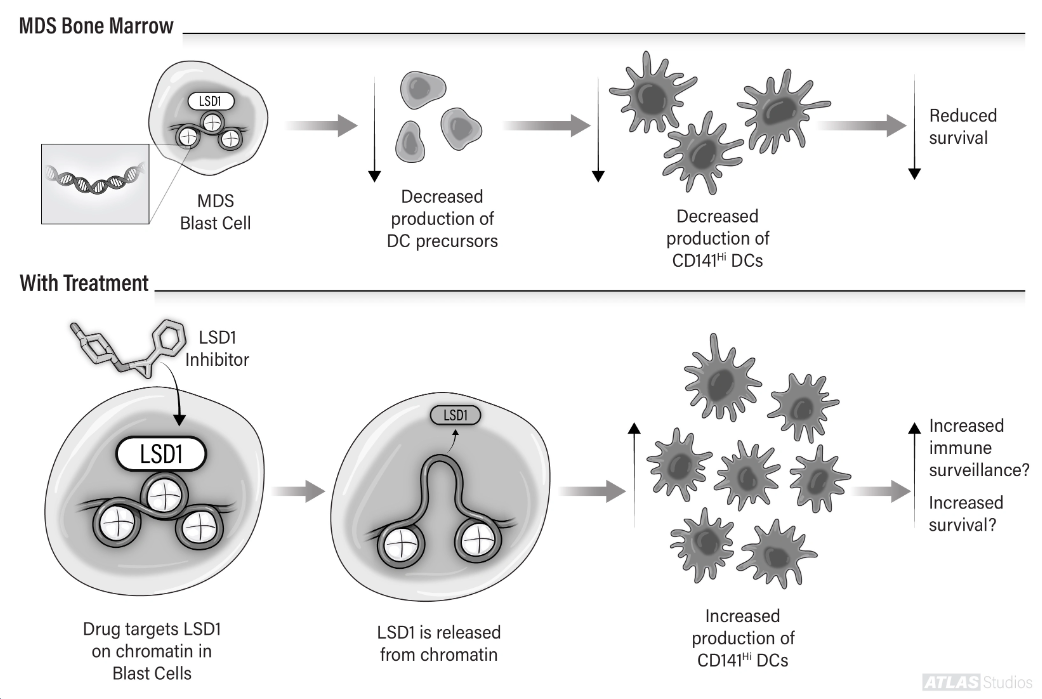
MDS clinical trials
Elizabeth Griffiths, MD, director of Roswell Park Comprehensive Cancer Center’s MDS patient services, works closely with Dr. Nemeth as she leads important clinical trials for emerging therapies and vaccine studies.
Clinical trial: NCT03358719
DEC-205/NY-ESO-1 Fusion Protein CDX-1401, Poly ICLC, Decitabine, and Nivolumab in Treating Patients with Myelodysplastic Syndrome or Acute Myeloid Leukemia
Clinical trial: NCT04484532
Evaluation of Antibody Response to High-Dose Seasonal Influenza Vaccination in Patients with Myeloid Malignancy Receiving Chemotherapy and Healthy Volunteers
Research focus: Triple negative breast cancer
With funding through a Breakthrough Award from the Department of Defense (DoD), Dr. Nemeth and Roswell Park colleague Scott Abrams, PhD, aim to improve the efficacy of immune checkpoint inhibitors by eliminating the production of myeloid-derived suppressor cells (MDSCs).
TNBC researchDoD research highlights
Webinar: New approaches with immunotherapies and breast cancer
“Tempering the Hostile Tumor Microenvironment to Boost Immunotherapy Outcomes in Triple-Negative Breast Cancer”
Dr. Nemeth and Dr. Abrams talked with the Breast Cancer Alliance and Focused Ultrasound Foundation about their work in understanding the processes that limit the efficacy of immune checkpoint inhibitors.
Publications
Research from the Nemeth Lab has been published in numerous influential journals.
Faculty affiliations
In the news
Connect with the Nemeth Lab
Email: Michael.Nemeth@RoswellPark.org
Phone: 716-845-1775
Department of Immunology
Roswell Park Comprehensive Cancer Center
Elm and Carlton Streets
Buffalo, NY 14263