Targeting metabolic shifting in breast cancer bone metastases
Current treatments for bone metastasis, such as bisphosphonates, have shown limited success. Such treatments predominantly target bone turnover and remodeling to ease complications instead of directly eradicating cancer cells in bone.
Subsequently, tumor cells that survive adjuvant therapies can eventually cause late-onset relapse in bone. Therefore, new therapeutic strategies are needed that can kill these cancer cells and improve the outcomes of patients with bone metastases.
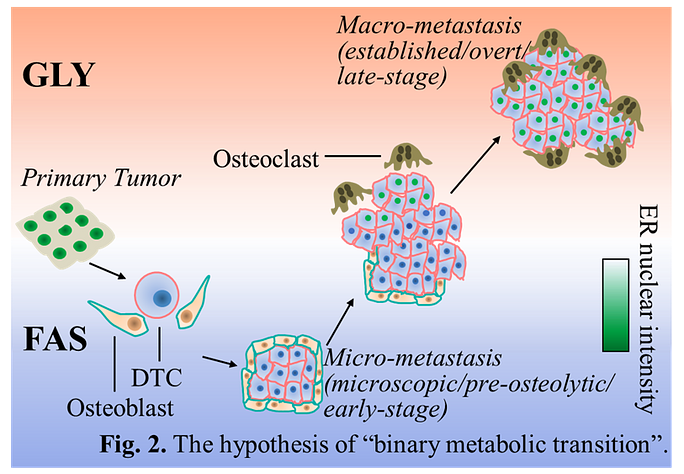
Recently, we have discovered that microscopic bone metastases grow slowly and undergo lipid synthesis during the early colonization stage to accumulate energy reserves, like an electronic device in energy-saver mode.
Then, as lesions gradually increase in size, the dominant metabolic program transitions to glycolysis, which allows cells to quickly break down glucose and extract the high amounts of energy necessary for proliferation.
While both lipid and glucose metabolism are attracting increasing attention in the field of cancer research, these pathways have not yet been fully exploited as druggable targets for breast cancer. One barrier might be the lack of understanding of whether these pathways represent true vulnerabilities.
In animal models, we will disrupt lipid and glucose metabolism at appropriate therapeutic windows to determine if this strategy can impede not only cancer growth in bone, but also its subsequent spread to other organs.
Completion of this project will establish the feasibility of this new treatment strategy and expediate timely translation of these findings into clinical practice.
Read the research
- Bado IL, et al. The bone microenvironment increases phenotypic plasticity of ER+ breast cancer cells. Dev Cell. 2021 Apr 19;56(8):1100-1117.e9. doi: 10.1016/j.devcel.2021.03.008.
- Muscarella AM, et al. Unique cellular protrusions mediate breast cancer cell migration by tethering to osteogenic cells. NPJ Breast Cancer. 2020 Sep 7;6:42. doi: 10.1038/s41523-020-00183-8.
- Kim IS, et al. Immuno-subtyping of breast cancer reveals distinct myeloid cell profiles and immunotherapy resistance mechanisms. Nat Cell Biol. 2019 Sep;21(9):1113-1126. doi: 10.1038/s41556-019-0373-7. Epub 2019 Aug 26.
- Wang H, Yu C, Zhang XH. Devil's Wake: Early-stage bone colonization by breast cancer. Mol Cell Oncol. 2015 Apr 14;3(3):e1026526. doi: 10.1080/23723556.2015.1026526.
- Wang H, et al. The osteogenic niche promotes early-stage bone colonization of disseminated breast cancer cells Cancer Cell. 2015 Feb 9;27(2):193-210. doi: 10.1016/j.ccell.2014.11.017. Epub 2015 Jan 15.
Contact the Wang Lab
Email: Hai.Wang@RoswellPark.org
Lab Location: Medical Research Complex - Room 358
Department of Molecular and Cellular Biology
Roswell Park Comprehensive Cancer Center
Elm and Carlton Streets
Buffalo, NY 14263