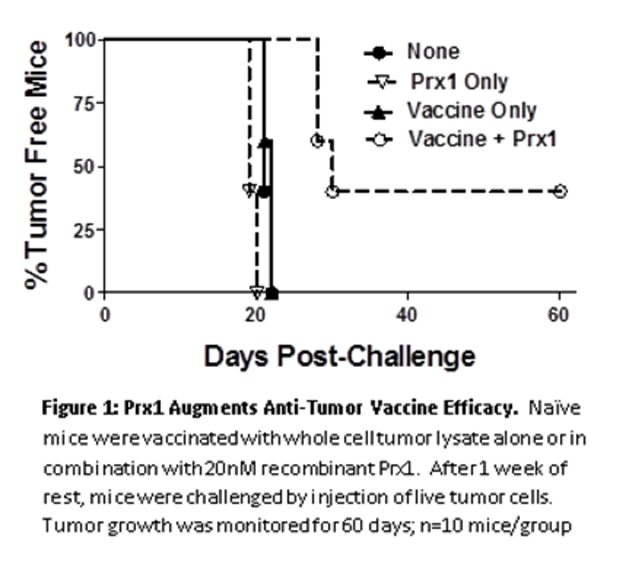The antioxidant Peroxiredoxin 1 (Prx1) is a molecular chaperone that acts as a regulator of hydrogen peroxide signaling, is overexpressed in many cancers, and is often secreted from tumor cells. Prx1 expression is elevated in various cancers, including esophageal, pancreatic, lung, follicular thyroid, and oral cancer. Elevated Prx1 levels have been linked with poor clinical outcomes and diminished overall patient survival. Recent studies have also shown that Prx1 can be secreted by non-small cell lung cancer cells. The function of secreted Prx1 is unknown and has not been previously exploited for therapeutic purposes.
Researchers at the Roswell Park Comprehensive Cancer Center have demonstrated that the inclusion of Prx1 with a vaccination to tumor antigens elicited macrophages or immature bone marrow-derived dendritic cells resulted in TLR4- dependent secretion of TNF-α and IL-6 and dendritic cell maturation leading to a more robust immune response.
Like other chaperone proteins and molecules involved in the innate immune response, PRrx1 is thought to play a role in the elicitation of robust, adaptive immune responses. The addition of Prx1 as a component of the vaccine adjuvant resulted in a marked increase in tumor free survival in a mouse model compared to a vaccine/adjuvant lacking Prx1.
Vaccinating against tumor antigens has thus far proven to not be a robust strategy for treating/preventing cancer. While there may be a multitude of reasons that this once-promising strategy has not worked, there is a belief that a priming of the innate immune system may be needed or helpful to elicit sustained immune response.