Treatment is another option for patients with refractory or relapsed large B-cell lymphoma
- Roswell Park is an authorized treatment center for Yescarta
- A select number of cancer centers across nation can administer Yescarta
- FDA-approved treatment is a form of CAR T-cell immunotherapy
BUFFALO, N.Y. — Roswell Park Comprehensive Cancer Center has been officially approved to administer an FDA-approved CAR T-cell therapy, Yescarta, to lymphoma patients. It’s manufactured by Kite, a Gilead Company.
Yescarta, also known as axicabtagene ciloleucel, is used to treat adult patients with diffuse large B-cell lymphoma (DLBCL), the most common type of non-Hodgkin Lymphoma. The immunotherapy treatment is created using a patient’s immune system to help fight the lymphoma. The patient’s T cells, a type of white blood cell, are collected and genetically modified to include a new gene that targets and kills the lymphoma cells. Once the cells are modified, they are infused back into the patient.
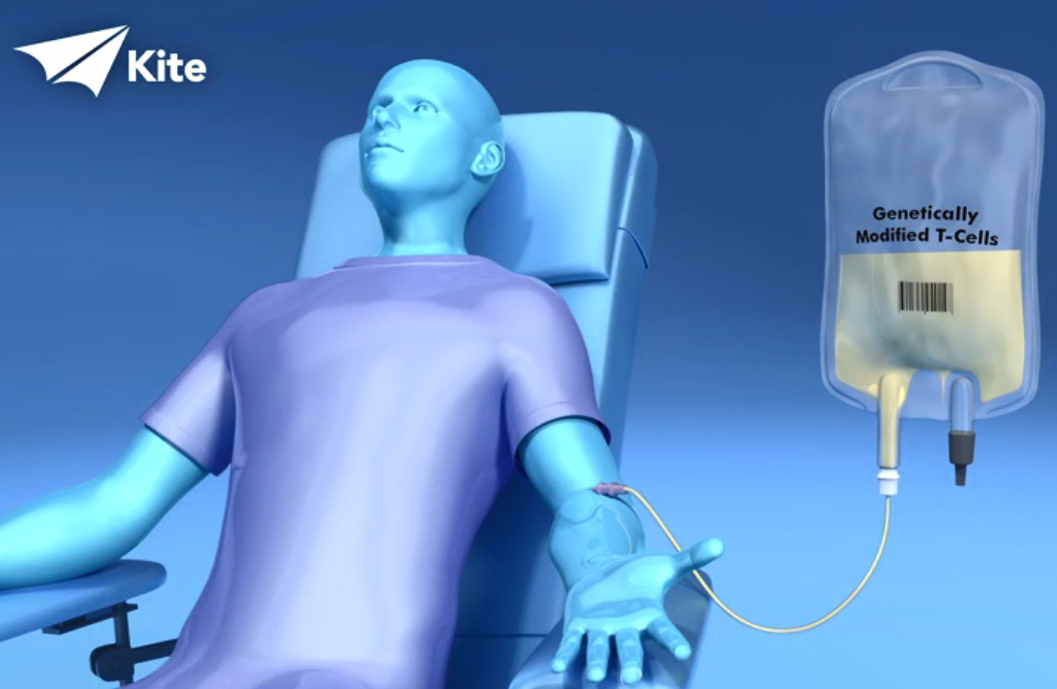
While chemotherapy is currently the standard way to treat the blood cancer, when it initially arises, doctors at Roswell Park can give Yescarta to adults with refractory or relapsed large B-cell lymphoma – those whose disease persists or returns despite treatment with chemotherapy.
“If chemotherapy isn’t working, that can be scary for a patient and their family. Now we have another treatment that has fewer side effects than chemotherapy and has the potential to stop the disease. It’s an exciting time,” says Dr. Francisco Hernandez-Ilizaliturri, MD, the Chief of Lymphoma and Myeloma at Roswell Park.
“Cellular and gene therapy is going to give hope to lymphoma patients who’ve tried chemotherapy, and it didn’t work for them. Innovative Chimeric Antigen Receptor (CAR) T-cell therapies tap into the patient’s immune system to attack the disease. This customized approach gives us a major tool to fight diffuse large B-cell lymphoma,” says Philip McCarthy, MD, Director of the Blood and Marrow Transplant Center and Professor of Oncology and Internal Medicine within the Department of Medicine at Roswell Park.
The cancer center was officially approved to administer the treatment to patients on February 28, 2018.
“Upon referral to Roswell Park, our doctors will assess each patient to determine whether this is an appropriate course of treatment. If so, members of our care team, from physicians to nurses are prepared to administer Yescarta,” says Steven Schinnagel, Ambulatory Center Administrator, in the Blood and Marrow Transplant Center.
Roswell Park Comprehensive Cancer Center is one of more than 30 hospitals across the nation that’s authorized to treat patients with Yescarta.
For more information about Yescarta, please visit: https://www.roswellpark.org/immunotherapy/about-immunotherapy/yescarta
###
Roswell Park Comprehensive Cancer Center is a community united by the drive to eliminate cancer’s grip on humanity by unlocking its secrets through personalized approaches and unleashing the healing power of hope. Founded by Dr. Roswell Park in 1898, it is the only National Cancer Institute-designated comprehensive cancer center in Upstate New York. Learn more at www.roswellpark.org, or contact us at 1-800-ROSWELL (1-800-767-9355) or ASKRoswell@roswellpark.org.
Annie Deck-Miller, Senior Media Relations Manager
716-845-8593; annie.deck-miller@roswellpark.org