The role of quiescent cancer cells in cancer drug resistance
Disordered cell replication, or proliferation, is a central feature of both hematologic and solid malignancies. However, even in aggressive cancers, a significant proportion of cancer cells do not proliferate for extended periods of time before re-entering the cell cycle, a state known as cancer cell quiescence.
Because nearly all cancer therapies preferentially target proliferating cancer cells, we and others have found that quiescent cancer cells (QCCs) are resistant to many cancer therapies and form the seeds of relapse and recurrence.
Tracking and characterizing QCCs
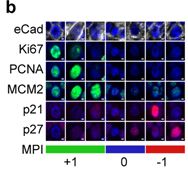
The Kabraji lab has a track record of characterizing and tracking treatment resistant QCCs in patient tissues and cancer models. We have shown that these QCCs can be found in premalignant ductal carcinoma in situ of the breast, in primary tumors of multiple types and in tumor metastases, using reproducible protein markers (e.g., AKTlow/HES1high).
While it was previously known that QCCs played a role in tumor spread and formation of metastatic disease, we have shown that QCCs within primary tumors promote tumor growth and represent a potential therapeutic target.
Multivariate proliferation index (MPI)
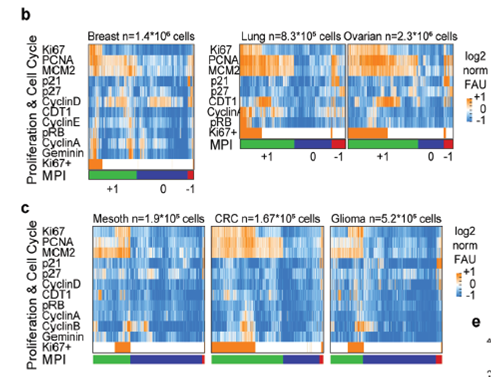
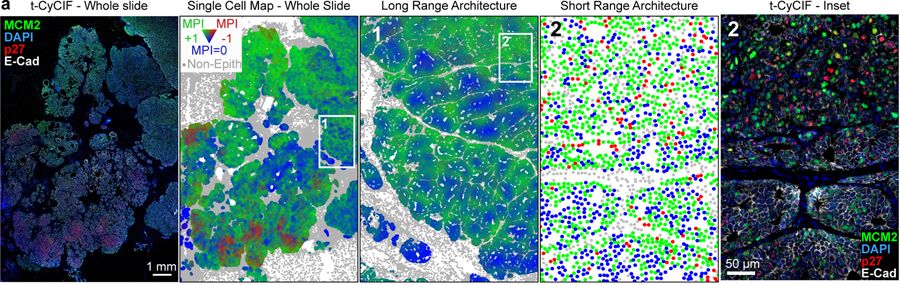
Using highly multiplexed immunofluorescence and whole slide tissue imaging technology known as Cyclic Immunofluorescence (CyCIF), we have developed a robust method to map and track QCCs in patient and model tissues: the Multivariate Proliferation Index (MPI).
Using this approach, we reported in Nature Cell Biology that proliferating and quiescent cancer cells exist in ‘neighborhoods’ adjacent or excluding specific immune cell subtypes.
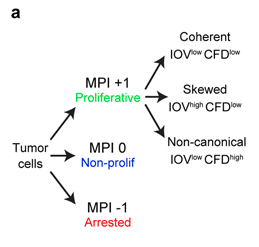
An example of tumor cells from a human breast cancer classified by the multivariate proliferation index (MPI) into proliferating (+1), quiescent (0) and arrested (-1) states.
MPI based classification allows assessment of cell cycle dynamics (coherence, skewed, non-canonical) in proliferating cells, allowing single cell analysis of response to perturbation and tissue geography.
We further reported that QCCs are enriched in residual disease after HER2 therapy in both patients and a genetically engineered mouse model, corroborating previous findings that QCCs are enriched after chemotherapy treatment in patients with triple negative breast cancer.
Ongoing research efforts
- Identifying the molecular programs that QCCs use to promote residual disease after HER2 therapy
- Characterizing QCC heterogeneity after drug treatment, metabolic and DNA damage stress
- Understanding how QCC subtypes interact with the immune system in primary and metastatic tumors
- Developing next-generation tissue biomarkers of drug resistance and response using tissues from patients
Contact the Kabraji Lab
Email: Sheheryar.Kabraji@RoswellPark.org
Office phone: 716-845-3429
Admin fax: 716-845-4620
Office location: Medical Research Complex - Room 458
Department of Molecular & Cellular Biology
Roswell Park Comprehensive Cancer Center
Elm and Carlton Streets
Buffalo, NY 14263