Methyl-H3K9 binding protein MPP8 promotes TSG silencing and EMT
H3K9 methylation is one of essential epigenetic marks which has been linked to a variety of biological processes including position-effect variegation, heterochromatin formation and transcriptional regulation.
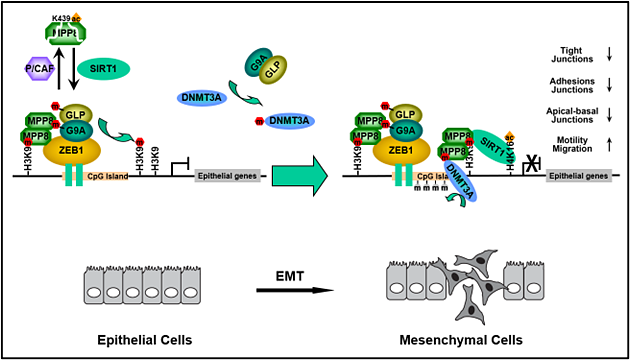
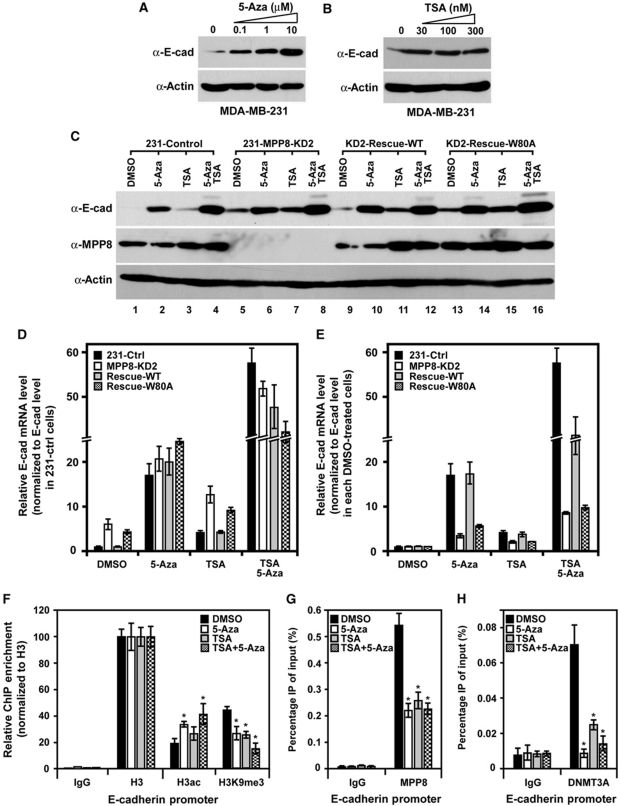
To better understand the function of H3K9 methylation, we have identified and characterized a novel methylated-H3K9 binding protein coined as MPP8 and revealed its pivotal roles in tumor suppressor gene (TSG) silencing and epithelial-mesenchymal transition (EMT) by coordinating histone H3K9 methylation and de novo DNA methylation on target promoters.
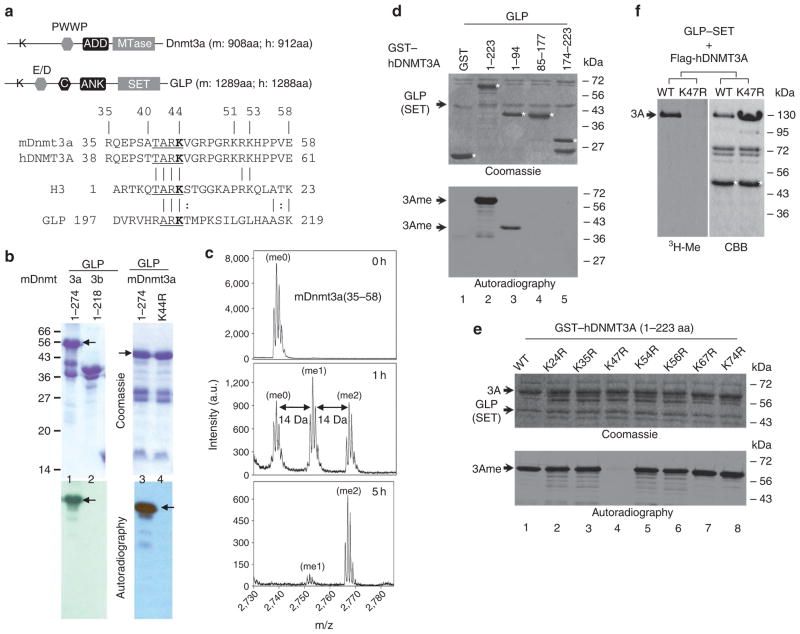
MPP8 also interacts with DNA methyltransferase 3A (DNMT3A), and histone H3K9 methyltransferases G9A and GLP.
We further uncovered that DNMT3A is methylated at lysine-47 (K47) by G9A and GLP which are also auto-methylated at lysine-185 (K185) and lysine-205 (K205) in their N-termini. Intriguingly, all these methylated-lysines are recognized by MPP8.
The methyl-H3K9 binding chromodomain of MPP8 forms a dimer both in solution and in crystals, suggesting that a dimeric MPP8 molecule could bridge the methylated DNMT3A and G9A/GLP, resulting in a repressive trimeric complex of DNMT3A-MPP8-G9A/GLP on chromatin templates.
These findings also provide a molecular explanation, at least in part, for the co-occurrence of DNA methylation and H3K9 methylation in chromatin.
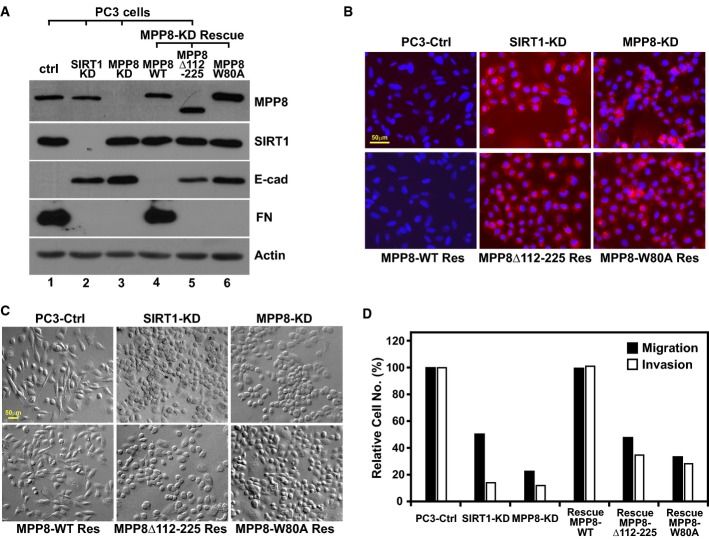
Moreover, we revealed that MPP8 crosstalks with histone deacetylase SIRT1 at multiple molecular layers - MPP8 recruits SIRT1 to deacetylate H4K16 for transcriptional repression while SIRT1 removes K439 acetylation on MPP8 to increase its protein stability.
We continue to investigate the functional implications of these novel molecular mechanisms in this complex epigenetic network coordinated by MPP8 in tumor formation, growth and metastasis.
See the science
MPP8 mediates the interactions between DNA methyltransferase Dnmt3a and H3K9 methyltransferase GLP/G9a
- Authors: Chang Y, Sun L, Kokura K, Horton JR, Fukuda M, Espejo A, Izumi V, Koomen JM, Bedford MT, Zhang X, Shinkai Y, Fang J, Cheng X
- Publication: Nat Commun. 2011 Nov 15:2:533
- DOI: 10.1038/emboj.2010.239
Connect with the Fang Lab
Department of Pharmacology and Therapeutics
Roswell Park Comprehensive Cancer Center
Elm and Carlton Streets
Buffalo, NY 14263